|
|
Service of microscopy.
In the Service of Microscopy of the COMAV Institute we offer to the scientific community (both public research institutions and private companies), the possibility of performing microscopic studies in any type of plant sample.
For this, we have a broad experience in the application of a wide range of microscopic techniques, together with all the equipment necessary to apply them to any type of plant sample. Among other techniques, we work with:
- Light microscopy (histology)
- Fluorescence microscopy
- Confocal laser scanning microscopy (CLSM)
- Transmission electron microscopy (TEM)
- Scanning electron microscopy (SEM)
- Low temperature sample processing (cryomethods) for transmission electron microscopy, including High Pressure Freezing (HPF) and Freeze Substitution (FS)
- Histochemical and cytochemical staining
- Immunofluorescence
- Inmunolocalization and quantification of immunogold labeling
- In situ hybridization
- Three-dimensional reconstruction for confocal and transmission electron microscopy
Contact.
José María Seguí Simarro.
e-mail: seguisim@btc.upv.es
Phone: (+34)963879047
Examples of our works
|
|
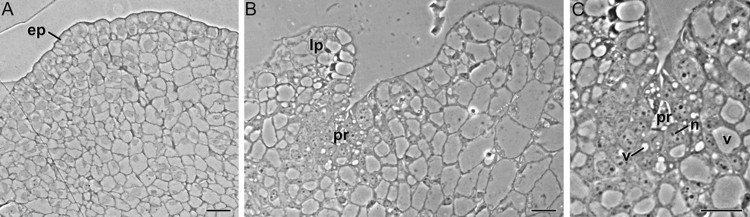
Histological study of the formation of adventitious buds from corm-derived calli in saffron (Crocus sativus). A: Section of a non-proliferating callus region. B: Proliferating region (pr) adjacent to a leaf primordium (lp). C: Detail of the proliferating region. Reference.
|
Top
|
|
|
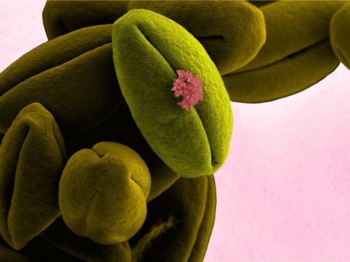
Tomato (Solanum lycopersicum) pollen samples observed by scanning electron microscopy and software-applied pseudocolor.
|
Top
|
|
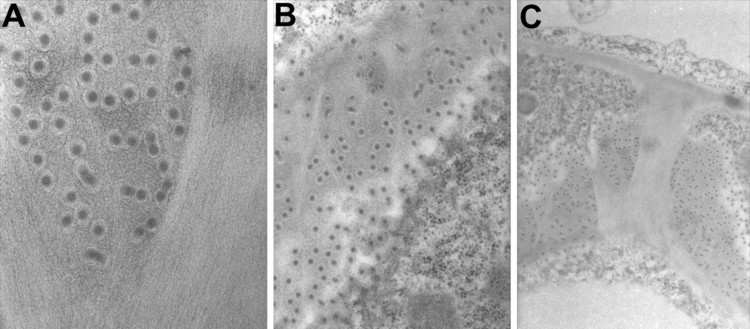
Ultrastructural detection by transmission electron microscopy of viroids of the Melon Necrotic Spot Virus (MNSV) in chemically fixed melon (Cucumis melo) samples.
|
Top
|
|
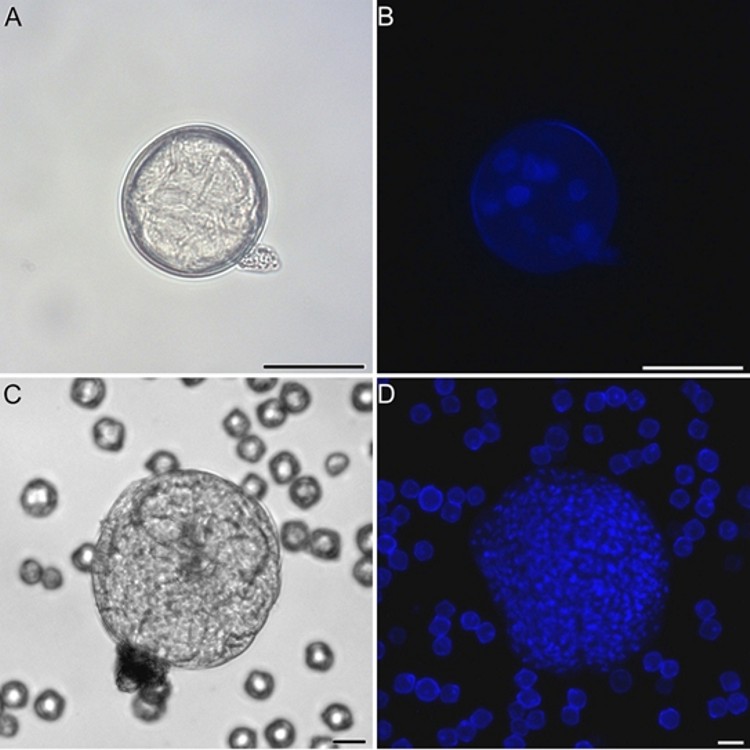
Eggplant (Solanum melongena) embryogenic microspores (A, B) and microspore-derived embryos (C, D) observed under phase contrast light microscopy (A, C) and fluorescence microscopy after DNA staining with DAPI (B, D).
|
Top
|
|
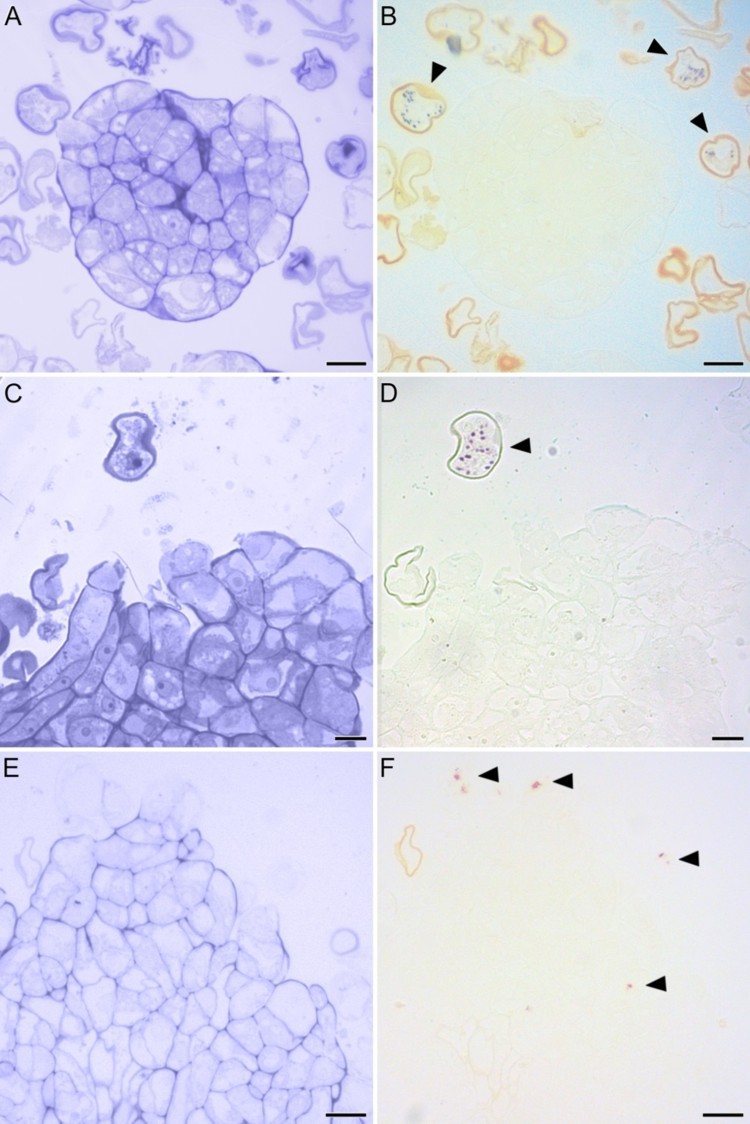
Eggplant (Solanum melongena) globular embryo derived from microspores, stained with Toluidine Blue (A) for protein and nucleic acid staining and Potassium Iodide-Iodine (IIK, B) for starch detection. Transitional embryo stained with Toluidine Blue (C) and IIK (D); callus stained with Toluidine Blue (E) and IIK (F). Arrowheads indicate the starch granules.
|
Top

|
|
Rapeseed (Brassica napus) seed observed under bright field light microscopy (A) and stained with Sudan Black for lipid detection (B).
|
Top

|
|
Three-dimensional model of a subcellular volume of a rapeseed (Brassica napus) embryogenic microspore developed from serial images of high pressure frozen, freeze-substituted samples, observed in a Field Emission Scanning Electron Microscope (FESEM) equipped with a Focused gallium Ion Beam (FIB). (A) Modeled subcellular volume. (B) Model excluding all the cell structures but the plastids (pl). The different plastid types are modeled in different colors: conventional (light green), open profiles engulfing cytoplasm (dark green), and closed profiles (yellow) with the engulfed cytoplasm (white). (C) Conventional, round-shaped plastid. (D) Disc-shaped plastid with a slight central depression (arrow). (E) Plastid starting to engulf cytoplasm. The arrow points to a deep depression that creates a cytoplasmic pocket within the plastid. (F,F') Cytoplasm-containing plastid where the internal cytoplasm is connected with the outer cytoplasm just by a narrow channel that ends in a small pore at the plastid surface. (F') 90° turn of this plastid, for a clear visualization of the narrow channel. Arrows point to the pore in (F) and to the narrow channel in (F'). (G) Round plastid (yellow) with the cytoplasmic contents (white) entirely isolated from the outer cytoplasm. cw, cell wall; m, mitochondrion; n, nucleus. Reference.
|
Top
|
|
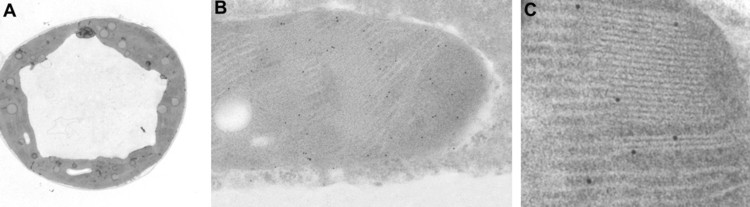
Ultrastructural localization of L1 proteins in chloroplasts of transgenic tobacco leaves.
|
Top

|
|
Ultrastructure of samples of embryo sacs (left), and cytoplasm (center) and nucleus (right) of rapeseed (Brassica napus) embryos cryofixed by high pressure freezing (HPF), processed by freeze-substitution, embedded in epoxy resin, and observed by transmission electron microscopy.
|
Top
|
|
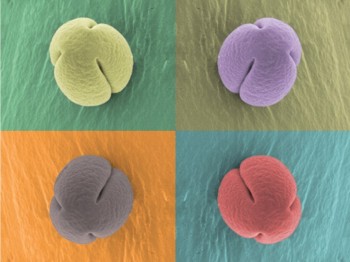
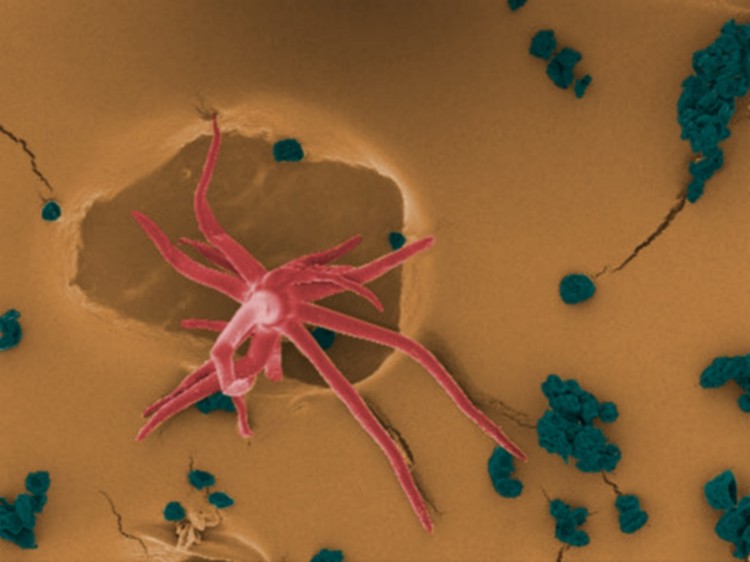
Eggplant (Solanum melongena) trichome (red) and pollen (green) observed by scanning electron microscopy and software-applied pseudocolor.
|
Top

|
|
|
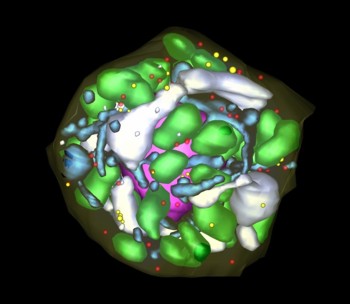
Interphase: G1
|
Interphase: G2
|
|
|
|
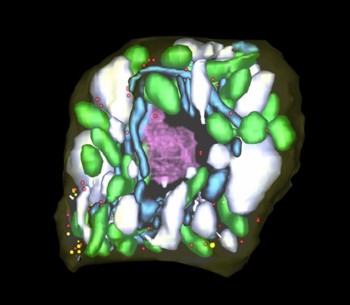
Mitosis: prophase
|
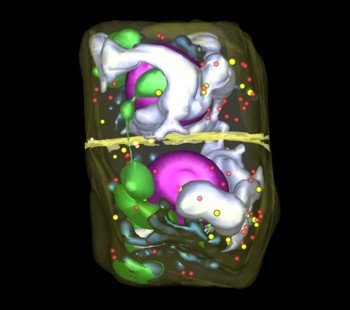
Mitosis: telophase + cytokinesis
|
|
|
Three-dimensional reconstruction of Arabidopsis thaliana meristematic cells at different stages of the cell cycle. Reference 1. Reference 2. Reference 3.
|
Top
|